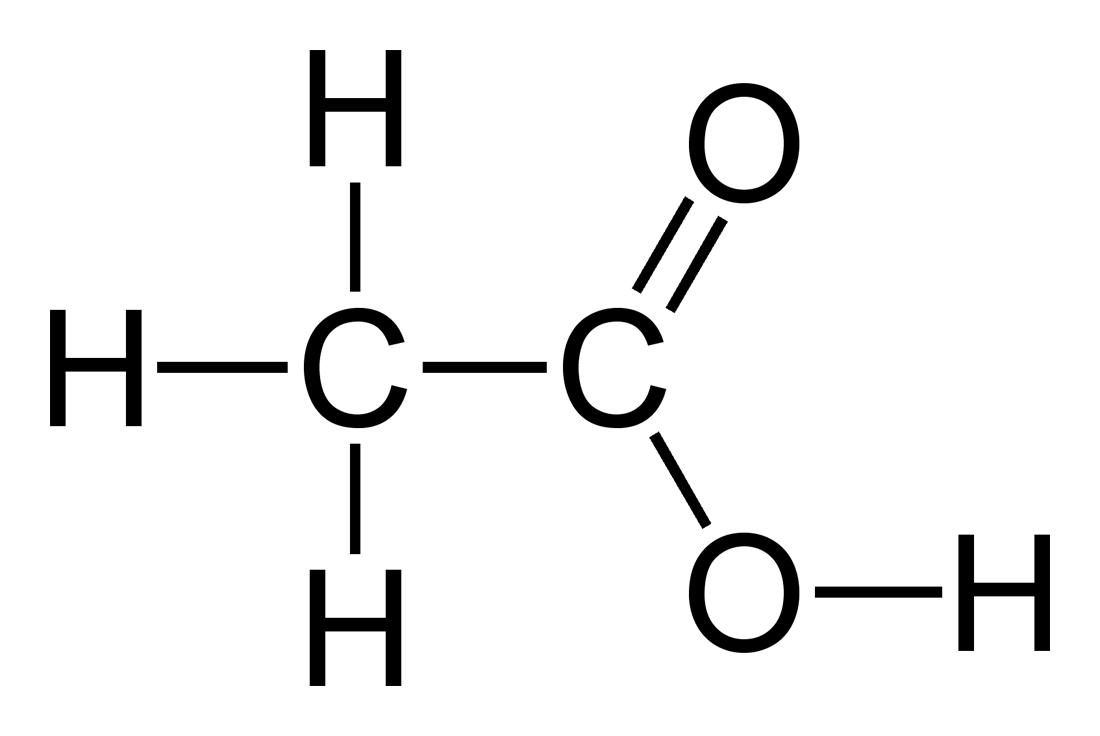
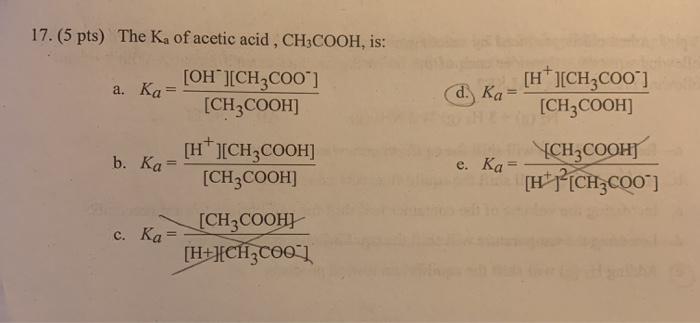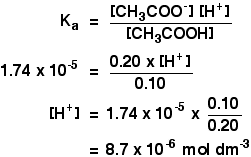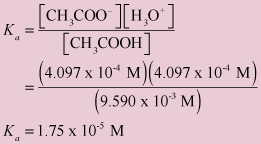A 0 05 n sol of acetic acid is found to be 1 9+ ionised at 25 c calculate - Chemistry - Equilibrium - 13276973 | Meritnation.com

A student prepared 0.10M acetic acid solution and experimentally measured its pH to be 2.88. Calculate Ka - Brainly.in
The pH of an acetic acid solution is 3.26. What is the concentration of acetic acid and what is the percent of acid that's ionized? - Quora
![What is the pH of a 1 M CH3COOH solution? [ Ka of acetic acid = 1.8 × 10^-5, Kw = 10^-14 mol^2 litre^-2 ] What is the pH of a 1 M CH3COOH solution? [ Ka of acetic acid = 1.8 × 10^-5, Kw = 10^-14 mol^2 litre^-2 ]](https://i.ytimg.com/vi/5MXjDjLyUp4/maxresdefault.jpg)
What is the pH of a 1 M CH3COOH solution? [ Ka of acetic acid = 1.8 × 10^-5, Kw = 10^-14 mol^2 litre^-2 ]
![The pH of 0.1 M acetic acid solution is closest to[Dissociation constant of the acid, Ka = 1.8 × 10^-5 ] The pH of 0.1 M acetic acid solution is closest to[Dissociation constant of the acid, Ka = 1.8 × 10^-5 ]](https://i.ytimg.com/vi/AufT6_CoFWY/maxresdefault.jpg)
The pH of 0.1 M acetic acid solution is closest to[Dissociation constant of the acid, Ka = 1.8 × 10^-5 ]